 This is the first in a series of posts under the general heading, “Whatever Happened to … ?” The idea is to revisit issues and opportunities of the past and help bring you up to date on what has been accomplished and what is still yet to be done. So … whatever happened to acid rain?
This is the first in a series of posts under the general heading, “Whatever Happened to … ?” The idea is to revisit issues and opportunities of the past and help bring you up to date on what has been accomplished and what is still yet to be done. So … whatever happened to acid rain?
As it pertains to the prepainted metal market, and especially to metal roofing, acid rain became an accepted and understood phenomenon in the late 1970s and early 1980s. Badly corroded roofs were becoming more prevalent, but—like any new problem—it took some time to identify the root cause and to learn how to overcome the issue of red rusting on roofs. And, as with any root cause analysis, an early root cause declaration is usually challenged. Then more research is done, more challenges are raised, and, eventually, consensus is reached.
Here’s the short story on acid rain: Coal-burning power plants that produce our electricity also produce sulfur dioxide (SO2) as a combustion by-product. The SO2 works its way into the atmosphere, combines with water, and eventually becomes sulfuric acid (H2SO4). Rain carries the H2SO4 from the sky to the ground. But what were we to do about this corrosion issue, and why is this happening now (thinking back to the 70s and 80s)? First the “Why now?” question.
As you can see in the graph below, SO2 has been with us for a while—since the beginning of the Industrial Revolution—but its prevalence grew at a furious rate beginning in the 1960s.

“Historical Sulfur Dioxide Emissions 1850-2000: Methods and Results,” S.J. Smith, E. Conception, R. Andres*, J. Lurz (2004). Pacific Northwest National Laboratory Report, PNNL–14537. *Department of Space Studies, University of North Dakota.
The growth in atmospheric SO2 concentration created a real problem for our industry. A common coil-coated substrate for the metal building industry back then—and, in fact, still today—was hot-dipped galvanized steel (HDG). Galvanizing is the process of dipping fabricated steel into bath of molten zinc.
Zinc is very reactive in an acid environment. As the zinc reacts with H2SO4, this protective galvanized metallic layer becomes thinner and thinner. By the way, the zinc is doing its job, which is to protect the steel substrate from rusting. This method of avoiding corrosion—where the zinc preferentially corrodes instead of the steel—is called “sacrificial corrosion protection” (i.e., the zinc is sacrificed in order to save the steel.) But there is only so much sacrificing that can be expected from a thin layer of zinc. When the zinc layer is sufficiently compromised, red rusting of the steel substrate occurs.
Another term that became common in the 1980s was the “acid rain belt.” Based first on observations and then on measurements, it was observed that a swath of the eastern U.S. had the worst acid rain problem. Here’s a mapped view from 1985: the red areas are those with the worst acid rain conditions (plotted here as H+ ion concentration, which is the acid component in acid rain).
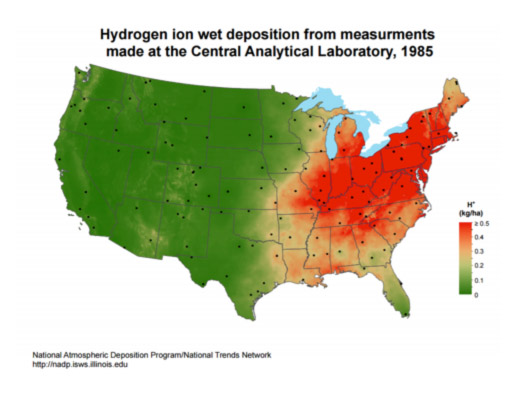
Why this region and not the West or the Plains? There are many complex variables at play, but let’s keep it simple at this point and look at how electricity is generated across our country. The following two maps show a very clear picture.
| Coal-Burning Power Plants, 2016
(produce 40% of all electricity in the U.S.) |
Natural Gas Power Plants, 2016
(produce 30% of all electricity in the U.S.) |
 |
 |
| https://www.eia.gov/state/maps.php | |
It is easy to see that not much coal is burned in the West. The primary fuel for producing electricity in the western U.S. is natural gas, which does not produce SO2. (Sadly, it—like coal, gasoline, and many other materials when burned or decomposed—produces carbon dioxide (CO2) a greenhouse gas, but let’s save that discussion for a future post.) So we now understand the “Why now?” and the “Why only in certain portions of the U.S.?” Knowledge is nice, but we had a problem to solve.
In an effort to overcome the rusting roofs issue, the coil coating industry tried a variety of approaches. One example: to protect the zinc, whose sole job is to protect the steel, a zinc phosphate pretreatment was used. (NOTE: Zinc phosphate was a common pretreatment in the earliest days of coil coating, but other pretreatments were developed to overcome some of the flexibility issues that were encountered in those early days. As it turns out, many of the worst flexibility issues had to do with a particular crystal form of the zinc metallic layer. But I digress; let’s leave zinc crystals for a future post.)
In the meantime, steel companies introduced Galvalume®, which, like HDG, is metallic-coated steel, but the protective layer is a blend of zinc and aluminum (at about a 50:50 ratio by weight). Aluminum is not nearly as reactive as zinc, and the metallic layer lasted longer than a pure zinc layer under acid rain conditions. Coatings companies worked to improve the flexibility of both their primers and topcoats in an effort to minimize cracking, since a crack is an open pathway for acid rain to reach the zinc layer. The combination of these improvements created significant performance advantages.
While the coil coating industry was doing everything it could to overcome the effects of acid rain, the EPA created the Clean Air Act of 1963. Whoa, 1963! Yes, it all began back then (which, by the way, is just one year after the birth of the National Coil Coating Association in 1962). The Clean Air Act grew to tackle many air-quality issues. Some complain mightily about EPA overreach, and certainly that is an issue, but look at the effect that the various regulations have had on the acid rain belt. (Compare this map from 2015 to the map from 1985 shown earlier in this post.)

We still live with the remnants of acid rain. Acidification of bodies of water has affected fish populations. Forest growth was another victim of acid rain. Trees recover, but it takes some time.
Do we at least have consensus about the origin of the problem? Google “acid rain hoax” and you’ll get thousands of hits (sometimes coupled with “myth of global warming”). Consensus is elusive, because causality can be endlessly debated. For me, the map above says it all. Acid rain has been ameliorated. And we continue to use many of the pretreatment and coating products formulated to combat this problem, which are now creating a superior barrier for any geographic environment.
David Cocuzzi, NCCA Technical Director
May 2017


April 24, 2019 at 3:44 pm
Hi Dave (NCCA). In light of the article from May 2017, we are still receiving complaints of drip-edge corrosion on roofs with painted (SMP and PVDF) Galvalume and HDG that is not very old. I would still say that acidity is a component in the edge corrosion. Installers can best prevent this in my experience by hemming the drip edge but most will not. The customer is generally not aware of this probability on a cut edge that holds moisture. The customer expects a 20-40 year roof with no aesthetic problems.
LikeLike